FDA Warns Using Homopathic Teething Tablets, Gel To Children
The US Food and Drug Association (FDA) has lately warned people to avoid over-the-counter natural treatment for teething problem of babies.
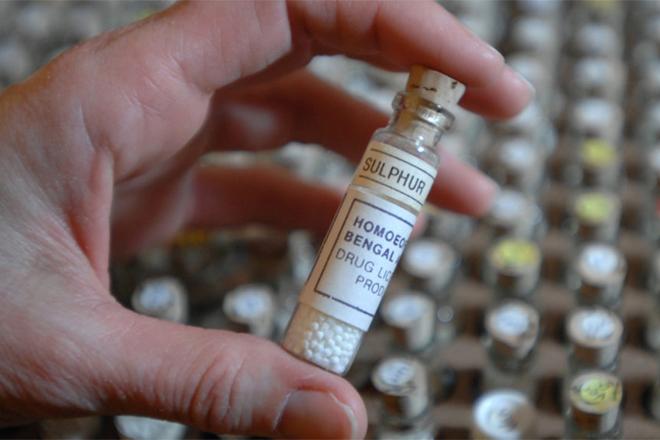
The agency has added consumers should not be using homeopathic teething tablets as those may pose a risk to infants and children.
FDA’s Center for Drug Evaluation and Research director Dr. Janet Woodcock said, “Teething can be managed without prescription or over-the-counter remedies… We recommend parents and caregivers not give homeopathic teething tablets and gels to children and seek advice from their health care professional for safe alternatives.”
The homeopathic teething tablets are distributed by CVS, Hyland’s, and are also found at several retail stores and online as well.
Presently the agency is investigating on the reports of adverse events linked to the homeopathic tablets as well as about the gels that include seizures in infants and children. It said such tablets and gels have not yet been approved for safety,
Earlier, about 6 years ago, the agency found Hyland’s Teething Tablets can cause serious harm if taken larger doses.
Parents are now urged strongly kids should be discontinue the tablets. The potential side effects of the tablets include seizures, difficulty breathing, lethargy, excessive sleepiness, muscle weakness, skin flushing, constipation, difficulty urinating, or agitation after using.